
The Melting point is 235 degrees Celsius and the boiling point is 178 degrees Celsius. Dichloro methane is also a polar solvent and it is less polar than water. 
These strong intermolecular forces make it difficult to break the molecule apart, therefore more energy is needed causing a high melting and boiling point. The structure of the compound caffeine is shown below: Water is a polar solvent. The London force is the immediate attraction of electrons from one atom to the positive nuclei of other surrounding atoms. 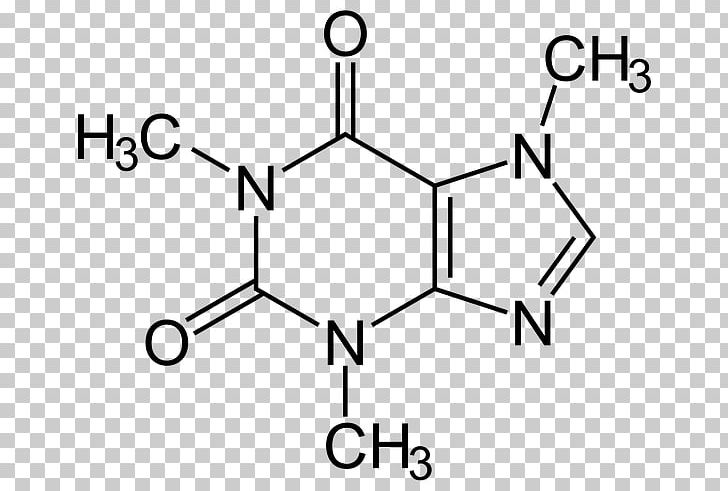
London Dispersion forces exist because the electrons are always in motion, temporarily becoming more positive or negative. These charged ends are attracted to other opposite charged ends creating a very strong intermolecular force. These bond dipoles within the molecule create an overall molecular dipole therefore the molecule has one negative end and one positive end. They occur whenever there is a separation of positive and negative charges. Caffeine molecules have bond dipoles that measure the polarity of a chemical bond within a molecule. Thus, caffeine is soluble in both water and polar organic solvents and significantly less soluble in non-polar solvents17. The intermolecular forces that are present in caffeine are London dispersion forces and Dipole-dipole forces.ĭipole-Dipole forces exist between the positive end of one polar molecule and the negative end of another polar molecule. The two carbonyl groups greatly add to the molecule's polarity along with the lone pair of electrons of the nitrogen. Caffeine is stopped by the membrane, because of its larger size relative to carbon dioxide, and collected.Intermolecular forces also known as van der Waals forces consist of Hydrogen bonding, London dispersion forces and Dipole-dipole forces. The carbon dioxide is then passed through a charcoal membrane that is selective toward carbon dioxide molecules. The resulting coffee beans are about 97% caffeine free. After this, they are immersed in supercritical carbon dioxide which binds to the caffeine molecules and draws them out of the beans, leaving the coffee solids (flavor) embedded in the bean. In order to use supercritical carbon dioxide to decaffeinate coffee beans, the beans are first steamed until they swell (this is where the co-solvent, water, comes into play). Water is more polar than caffeine is, so supercritical carbon dioxide, in the presence of a co-solvent like water, will dissolve the more non-polar substance, in this case, caffeine. Although supercritical carbon dioxide is non-polar, and should only be able to dissolve non-polar substances, certain co-solvents, like water, can be added so that supercritical carbon dioxide can actually dissolve polar molecules like caffeine. It behaves like gas, and permeates a porous substance, while also exhibiting liquid properties to dissolve substances. This method is repeated until the coffee beans are 99.9% decaffeinated, and the flavor is left intact.Ĭarbon dioxide supercritical fluid (temperature above 31.1 ☌ and pressure above 73 atm) exhibits both liquid and gas-like behavior. Since the only difference between the mixture and the fresh coffee beans is the caffeine concentration, caffeine molecules will diffuse out of the beans into the mixture of coffee solids, leaving the coffee beans caffeine free. Concentration gradients take advantage of the law of simple diffusion- the movement of molecules from an area of high solute concentration to an area of low solute concentration in order to 'even out' the uneven distribution of molucules. The mixture creates a concentration gradient when added to a fresh batch of coffee beans. After filtration, the mixture that is left is water saturated with coffee flavor molecules – referred as “coffee solids” by the manufacturers. The activated carbon filter has holes large enough to allow water and coffee solids (smaller molecules) to pass through, but not caffeine (relatively larger molecule). The carbon filter is made out of activated carbon, carbon that has been made porous through the process of carbonization (reacting carbon in anaerobic conditions until the gaps between carbon atoms are large enough to allow molecules to pass through). The beans are then discarded, and the solution of water, caffeine, and coffee solids is passed through a carbon filter. First, unroasted (green) coffee beans are soaked in water until caffeine is dissolved in water. The Swiss Water Processing method removes caffeine without using any chemicals, but instead applies the law of simple diffusion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
